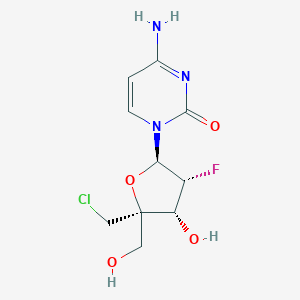
-
ALS-8112
- names:
ALS-8112
- CAS号:
1445379-92-9
MDL Number: No data available - MF(分子式): C10H13ClFN3O4 MW(分子量): 293.68
- EINECS:No data available Reaxys Number:No data available
- Pubchem ID:71621663 Brand:BIOFOUNT
| 货品编码 | 规格 | 纯度 | 价格 (¥) | 现价(¥) | 特价(¥) | 库存描述 | 数量 | 总计 (¥) |
|---|---|---|---|---|---|---|---|---|
| YZM000865-5mg | 5mg | 99.9% | ¥ 3540.00 | ¥ 3540.00 | 2-3天 | ¥ 0.00 | ||
| YZM000865-1mg | 1mg | 99.9% | ¥ 1462.00 | ¥ 1462.00 | 2-3天 | ¥ 0.00 |
| 中文别名 | ALS-8112(1445379-92-9);ALS8112;ALS 8112 |
| 英文别名 | ALS-8112,1445379-92-9 |
| CAS号 | 1445379-92-9 |
| Inchi | InChI=1S/C10H13ClFN3O4/c11-3-10(4-16)7(17)6(12)8(19-10)15-2-1-5(13)14-9(15)18/h1-2,6-8,16-17H,3-4H2,(H2,13,14,18)/t6-,7+,8-,10-/m1/s1 |
| InchiKey | AWSRKKBIPSQHOJ-IBCQBUCCSA-N |
| 分子式 Formula | C10H13ClFN3O4 |
| 分子量 Molecular Weight | 293.68 |
| 溶解度Solubility | 生物体外In Vitro:DMSO溶解度≥ 47 mg/mL(160.04 mM)*"≥" means soluble可溶, but saturation unknown溶解度未知. |
| 性状 | 白色至灰白色固体粉末 |
| 储藏条件 Storage conditions | -20°C 3 years年 4°C 2 years年 / 溶液中:-80°C 6 months月 -20°C 1 month月 |
ALS-8112(1445379-92-9)实验注意事项:
1.实验前需戴好防护眼镜,穿戴防护服和口罩,佩戴手套,避免与皮肤接触。
2.实验过程中如遇到有毒或者刺激性物质及有害物质产生,必要时实验操作需要手套箱内完成以免对实验人员造成伤害
3.实验后产生的废弃物需分类存储,并交于专业生物废气物处理公司处理,以免造成环境污染
ALS-8112(1445379-92-9) Experimental considerations:
1. Wear protective glasses, protective clothing and masks, gloves, and avoid contact with the skin during the experiment.
2. The waste generated after the experiment needs to be stored separately, and handed over to a professional biological waste gas treatment company to avoid environmental pollution.
Tag:ALS-8112(1445379-92-9),ALS-8112试剂,ALS-8112的纯度,ALS-8112抑制剂,ALS-8112的含量,ALS-8112的作用,ALS-8112的合成,ALS-8112的使用,ALS-8112的外观,ALS-8112的性质,ALS-8112的生产,ALS-8112的MSDS,ALS-8112的厂家
| 产品说明 | ALS-8112(1445379-92-9)是高效,选择性的呼吸道合胞病毒(RSV)聚合酶的抑制剂 |
| Introduction | ALS-8112(1445379-92-9) is a potent and selective respiratory syncytial virus (RSV) polymerase inhibitor. The 5'riphosphate form of ALS112 inhibits RSV polymerase with anIC50of 0.02 μM. |
| Application1 | |
| Application2 | |
| Application3 |
| 警示图 | |
| 危险性 | warning |
| 危险性警示 | Not available |
| 安全声明 | H303吞入可能有害+H313皮肤接触可能有害+H2413吸入可能对身体有害 |
| 安全防护 | P264处理后彻底清洗+P280戴防护手套/穿防护服/戴防护眼罩/戴防护面具+P305如果进入眼睛+P351用水小心冲洗几分钟+P338取出隐形眼镜(如果有)并且易于操作,继续冲洗+P337如果眼睛刺激持续+P2393获得医疗建议/护理 |
| 备注 | 实验过程中防止吸入、食入,做好安全防护 |
| Wang G, et al. Discovery of 4'-chloromethyl-2'-deoxy-3',5'-di-O-isobutyryl-2'-fluorocytidine (ALS-8176), a first-in-class RSV polymerase inhibitor for treatment of human respiratory syncytial virus in |
| Jordan PC, et al. Activation Pathway of a Nucleoside Analog Inhibiting Respiratory Syncytial Virus Polymerase. ACS Chem Biol. 2017 Jan 20;12(1):83-91. |
| DeVincenzo JP, et al. Activity of Oral ALS-008176 in a Respiratory Syncytial Virus Challenge Study. N Engl J Med. 2015 Nov 19;373(21):2048-58. |
| Deval J, et al. Molecular Basis for the Selective Inhibition of Respiratory Syncytial Virus RNA Polymerase by 2'-Fluoro-4'-Chloromethyl-Cytidine Triphosphate. PLoS Pathog. 2015 Jun 22;11(6):e1004995. |
Discovery of 4'-chloromethyl-2'-deoxy-3',5'-di-O-isobutyryl-2'-fluorocytidine (ALS-8176), a first-in-class RSV polymerase inhibitor for treatment of human respiratory syncytial virus infection
Abstract:Respiratory syncytial virus (RSV) is a leading pathogen of childhood and is associated with significant morbidity and mortality. To date, ribavirin is the only approved small molecule drug, which has limited use. The only other RSV drug is palivizumab, a monoclonal antibody, which is used for RSV prophylaxis. Clearly, there is an urgent need for small molecule RSV drugs. This article reports the design, synthesis, anti-RSV activity, metabolism, and pharmacokinetics of a series of 4'-substituted cytidine nucleosides. Among tested compounds 4'-chloromethyl-2'-deoxy-2'-fluorocytidine (2c) exhibited the most promising activity in the RSV replicon assay with an EC50 of 0.15 μM. The 5'-triphosphate of 2c (2c-TP) inhibited RSV polymerase with an IC50 of 0.02 μM without appreciable inhibition of human DNA and RNA polymerases at 100 μM. ALS-8176 (71), the 3',5'-di-O-isobutyryl prodrug of 2c, demonstrated good oral bioavailability and a high level of 2c-TP in vivo. Compound 71 is a first-in-class nucleoside RSV polymerase inhibitor that demonstrated excellent anti-RSV efficacy and safety in a phase 2 clinical RSV challenge study.
Activation Pathway of a Nucleoside Analog Inhibiting Respiratory Syncytial Virus Polymerase
Abstract:Human respiratory syncytial virus (RSV) is a negative-sense RNA virus and a significant cause of respiratory infection in infants and the elderly. No effective vaccines or antiviral therapies are available for the treatment of RSV. ALS-8176 is a first-in-class nucleoside prodrug inhibitor of RSV replication currently under clinical evaluation. ALS-8112, the parent molecule of ALS-8176, undergoes intracellular phosphorylation, yielding the active 5'-triphosphate metabolite. The host kinases responsible for this conversion are not known. Therefore, elucidation of the ALS-8112 activation pathway is key to further understanding its conversion mechanism, particularly given its potent antiviral effects. Here, we have identified the activation pathway of ALS-8112 and show it is unlike other antiviral cytidine analogs. The first step, driven by deoxycytidine kinase (dCK), is highly efficient, while the second step limits the formation of the active 5'-triphosphate species. ALS-8112 is a 2'- and 4'-modified nucleoside analog, prompting us to investigate dCK recognition of other 2'- and 4'-modified nucleosides. Our biochemical approach along with computational modeling contributes to an enhanced structure-activity profile for dCK. These results highlight an exciting potential to optimize nucleoside analogs based on the second activation step and increased attention toward nucleoside diphosphate and triphosphate prodrugs in drug discovery.
Activity of Oral ALS-008176 in a Respiratory Syncytial Virus Challenge Study
Abstract:BACKGROUND Respiratory syncytial virus (RSV) infection is a cause of substantial morbidity and mortality. There is no known effective therapy. METHODS We conducted a randomized, double-blind, clinical trial in healthy adults inoculated with RSV. Participants received the oral nucleoside analogue ALS-008176 or placebo 12 hours after confirmation of RSV infection or 6 days after inoculation. Treatment was administered every 12 hours for 5 days. Viral load, disease severity, resistance, and safety were measured throughout the 28-day study period, with measurement beginning before inoculation. The primary end point was the area under the curve (AUC) for viral load, which was assessed immediately before administration of the first dose through the 12th day after inoculation in participants infected with RSV. RESULTS A total of 62 participants received placebo or one of three ALS-008176 dosing regimens: 1 loading dose of 750 mg followed by 9 maintenance doses of 500 mg (group 1), 1 loading dose of 750 mg followed by 9 maintenance doses of 150 mg (group 2), or 10 doses of 375 mg (group 3). In the 35 infected participants (23 of whom were treated with ALS-008176), the AUCs for viral load for groups 1, 2, and 3 and the placebo group were 59.9, 73.7, 133.4, and 500.9 log10 plaque-forming-unit equivalents × hours per milliliter, respectively (P≤0.001). The time to nondetectability on polymerase-chain-reaction assay (P<0.001), the peak viral load (P≤0.001), the AUC for symptom score (P<0.05), and the AUC for mucus weight were lower in all groups receiving ALS-008176 than in the placebo group. Antiviral activity was greatest in the two groups that received a loading dose--viral clearance was accelerated (P≤0.05), and the AUC for viral load decreased by 85 to 88% as compared with the placebo group. Within this small trial, no viral rebound or resistance was identified. There were no serious adverse events, and there was no need for premature discontinuation of the study drug. CONCLUSIONS In this RSV challenge study, more rapid RSV clearance and a greater reduction of viral load, with accompanying improvements in the severity of clinical disease, were observed in the groups treated with ALS-008176 than in the placebo group. (Funded by Alios BioPharma; ClinicalTrials.gov number, NCT02094365.).
- 相关产品
-
< >
- 推荐产品
-
< >
- 最新产品
-
< >
新闻
怎么做细胞爬片免疫组化染色实验
细胞爬片免疫组化染色,是通过细胞爬片是让玻片浸在细胞培养基内,细胞在玻片上生长,主要用于组织学,免疫组织化学...
2020/7/20 22:04:33
提取病毒RNA的实验方法
提取病毒RNA方法分别有:异硫氰酸胍的提取病毒RNA方法、TRIzol LS提取法、Trizol法提取法等等...
2020/7/22 20:29:26

细胞培养耗材技术领先性
细胞培养板行业面临的核心痛点是:常规TC处理后表面亲水角随时间衰减,影响长期培养稳定性,BIOFOUNT高分...
2026/4/28 15:12:51
细胞培养耗材关键性能
细胞培养板的水接触角作为表面润湿性的核心指标,直接影响细胞贴壁、增殖、分化及功能表达,其中40°(低接触角/...
2026/4/28 14:52:44

chelex 100树脂国产替代之路-BIOFOUNT范德生物
Chelex 100螯合离子交换树脂对铜、铁和其他重金属?的偏好显著高于对钠、钾等一价阳离子的偏好。它对二价...
2025/11/4 14:22:46

9月开学季——助研新学期 范德送好礼
2025/8/28 15:30:55

Waxfilm 实验室封口膜:技术与国际市场的双重突破
在实验室耗材领域,封口膜是保障实验准确性与稳定性的关键产品之一。近年来,Waxfilm?实验室封口膜凭借其卓...
2025/5/13 13:03:40

Waxfilm实验室封口膜的5大突破
Waxfilm实验室封口膜作为生物功能膜领域的国产技术突破和品牌突破,是生物领域中国技术发展的缩影。
2025/5/6 17:02:07

各种微流控芯片键合方法的优缺点
微流控芯片键合:目前主要有激光焊接、热压键合、胶键合、超音波焊接,每种方法都有各自的优缺点。本文主要介绍聚酯...
2023/7/28 10:43:09

新一代微流控键合解决方案
微流控键合解决方案:微流控芯片制造的一个重要环节,也是最容易被忽视的--芯片键合。其中一个重要因素是:微流控...
2023/7/27 12:44:28



 购物车
购物车 



