-
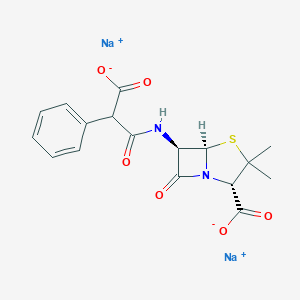
羧苄青霉素钠
NMR and HPLC COA下载 MSDS下载 - Names:
Carbenicillin disodium
- CAS号:
4800-94-6
MDL Number: MFCD00077683 - MF(分子式): C17H16N2O6SNa2 MW(分子量): 422.36
- EINECS:225-360-8 Reaxys Number:
- Pubchem ID: Brand:BIOFOUNT
| 货品编码 | 规格 | 纯度 | 价格 (¥) | 现价(¥) | 特价(¥) | 库存描述 | 数量 | 总计 (¥) |
|---|---|---|---|---|---|---|---|---|
| SS0406-25g | 25g | USP | ¥ 708.00 | ¥ 708.00 | 3-5days | ¥ 0.00 | ||
| SS0406-5g | 5g | USP | ¥ 236.00 | ¥ 236.00 | Instock | ¥ 0.00 | ||
| SS0406-1g | 1g | USP | ¥ 98.00 | ¥ 98.00 | Instock | ¥ 0.00 |
| 中文别名 | 羧苄青霉素钠,羧苄青霉素钠(4800-94-6,Carbenicillin disodium);羧苄基青霉素双钠盐;羧苄西林二钠盐;Α-羰基苄基青霉素二钠;羧苄青霉素二钠盐;羰苄青霉素钠 |
| 英文别名 | Carbenicillin disodium(4800-94-6);Carbenicillin disodium salt;Sodium carbenicillin |
| CAS号 | 4800-94-6 |
| Inchi | InChI=1S/C17H18N2O6S.2Na/c1-17(2)11(16(24)25)19-13(21)10(14(19)26-17)18-12(20)9(15(22)23)8-6-4-3-5-7-8;;/h3-7,9-11,14H,1-2H3,(H,18,20)(H,22,23)(H,24,25);;/q;2*+1/p-2/t9?,10-,11+,14-;;/m1../s1 |
| InchiKey | RTYJTGSCYUUYAL-YCAHSCEMSA-L |
| 分子式 Molecular Weight | C17H16N2O6SNa2 |
| 分子量 Formula | 422.36 |
| 溶解度Solubility | 略溶于DMSO,甲醇(超声) |
| 性状 | 浅黄色固体粉末 |
| 储藏条件 Storage conditions | 储存温度-20°C |
羧苄青霉素钠(4800-94-6,Carbenicillin disodium)毒理属性测试:
| 生物 | 测试类型 | 路线 | 报告剂量(标准化剂量) | 影响 | 参考 |
|---|---|---|---|---|---|
| rat | LD50 | oral | >10 gm/kg (10000 mg/kg) | Drugs in Japan, 6(187), 1982 | |
| rat | LD50 | intraperitoneal | 10 gm/kg (10000 mg/kg) | Drugs in Japan, 6(187), 1982 | |
| rat | LD50 | subcutaneous | >10 gm/kg (10000 mg/kg) | Drugs in Japan, 6(187), 1982 | |
| rat | LD50 | intravenous | 6800 mg/kg (6800 mg/kg) | Drugs in Japan, 6(187), 1982 | |
| mouse | LD50 | oral | >12 gm/kg (12000 mg/kg) | Chemotherapy, 23(572), 1975 |
羧苄青霉素钠(4800-94-6,Carbenicillin disodium)实验注意事项:
1.实验前需戴好防护眼镜,穿戴防护服和口罩,佩戴手套,避免与皮肤接触。
2.实验过程中如遇到有毒或者刺激性物质及有害物质产生,必要时实验操作需要手套箱内完成以免对实验人员造成伤害。
3.取样品的移液枪头需及时更换,必要时为避免交叉污染尽可能选择滤芯吸头。
4.称量药品时选用称量纸,并无风处取药和称量以免扬撒,试剂的容器使用前务必确保干净,并消毒。
5.取药品时尽量采用多个药勺分别使用,使用后清洗干净后,烘干消毒存放。
6.实验后产生的废弃物需分类存储,并交于专业生物废气物处理公司处理,以免造成环境污染。
Carbenicillin disodium(4800-94-6) Experimental considerations:
1. Wear protective glasses, protective clothing and masks, gloves, and avoid contact with the skin during the experiment.
2. The waste generated after the experiment needs to be stored separately, and handed over to a professional biological waste gas treatment company to avoid environmental pollution.
Tag:羧苄青霉素钠(4800-94-6,Carbenicillin disodium),羧苄青霉素钠试剂,羧苄青霉素钠的纯度,羧苄青霉素钠的储存,羧苄青霉素钠的外观,羧苄青霉素钠的溶解度,羧苄青霉素钠的厂家,羧苄青霉素钠的价格,羧苄青霉素钠的使用注意事项,羧苄青霉素钠的MSDS,羧苄青霉素钠的游离态
| 产品说明 | 羧苄青霉素钠(4800-94-6)仅做科学研究使用,羧苄青霉素钠MSDS,羧苄青霉素钠溶解度等见主页。 |
| Introduction | Carbenicillin sodium (4800-94-6,羧苄青霉素钠) is only used for scientific research and chemical synthesis intermediates. For other parameters of 4800-94-6, see the homepage |
| Application1 | 羧苄青霉素钠是一种源自青霉素的β-内酰胺抗生素,可抑制肽聚糖的合成,肽聚糖是细菌细胞壁的关键成分。 |
| Application2 | 依靠在细菌细胞膜的内层面使转肽酶(transpeptidases)失活抑制细菌细胞壁的合成 |
| Application3 | 类似于青霉素 |
| 警示图 | |
| 危险性 | warning |
| 危险性警示 | No data available |
| 安全声明 | H317,H334 |
| 安全防护 | P261,P280,P342+P311 |
| 备注 | 实验过程中防止吸入、食入,做好安全防护 |
| 象形图 |   |
|---|---|
| 信号 | Danger |
| GHS危险说明 |
Aggregated GHS information provided by 207 companies from 4 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies. H317 (99.52%): May cause an allergic skin reaction [Warning Sensitization, Skin] H334 (99.03%): May cause allergy or asthma symptoms or breathing difficulties if inhaled [Danger Sensitization, respiratory] Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown. |
| 防范说明代码 |
P261, P272, P280, P285, P302+P352, P304+P341, P321, P333+P313, P342+P311, P363, and P501 (The corresponding statement to each P-code can be found at the GHS Classification page.) |
| Influences of antibiotics on plantlet regeneration via organogenesis in loblolly pine (Pinus taeda L.)(Journal of Forestry Research,2003) |
| Induction of hairy roots by various strains of Agrobacterium rhizogenes in different types of Capsicum species explants(BMC Research Notes,2014) |
| Fc Engineering: Tailored Synthetic Human IgG1-Fc Repertoire for High-Affinity Interaction with FcRn at pH 6.0(Antibody Engineering,2018) |
| The functional effect of Gly209 and Ile213 substitutions on lysozyme activity of family 19 chitinase encoded by cyanophage Ma-LMM01(Fisheries Science,2011) |
| Cloning and analysis of a functional promoter of fungal immunomodulatory protein from Flammulina velutipes(Molecular Biology Reports,2014) |
1.Intropin (dopamine hydrochloride) intravenous admixture compatibility. Part 2: stability with some commonly used antibiotics in 5% dextrose injection.
Gardella LA;Kesler H;Carter JE;Zaroslinski JF Am J Hosp Pharm. 1976 Jun;33(6):537-40.
The stability of dopamine hydrochloride (Intropin) and several commonly used antibiotics was studied as admixtures in 5% Dextrose Injection USP. The antibiotic-dopamine-dextrose 5% admixtures were assayed for dopamine by colorimetric and chromatographic procedures. The antibiotics were assayed by standard microbiological methods. Kanamycin sulfate, tetracycline hydrochloride, carbenicillin disodium and chloramphenicol sodium succinate were stable in the Intropin-5% dextrose admixture for a period of 24 hours at room temperature in fluorescent and natural (western exposure) light. Gentamicin sulfate, penicillin G potassium and cephalothin sodium were stable in Intropin-5% dextrose admixture for six hours. Ampicillin sodium was stable in the Intropin admixture for only one hour. Amphotericin B was physically unstable in the Intropin-dextrose 5% solution upon admixture. The potency of dopamine hydrochloride remained substantially unchanged in the presence of the above antibiotics. It is recommended that dopamine not be added to amphotericin B or ampicillin sodium admixtures. Further, in order to avoid a fixed combination of potent drugs, it is recommended that a "piggyback" administration set or administration into a second injection site be employed when another drug is to be administered with dopamine hydrochloride.
2.Piperacillin and gentamicin v carbenicillin and gentamicin for treatment of serious gram-negative infections.
Kohler RB;Foerster LA;Wheat LJ;Williams JM;Reynolds J;Norton J Arch Intern Med. 1982 Jul;142(7):1335-7.
Piperacillin sodium, a new penicillin with remarkable in vitro activity against Pseudomonas aeruginosa and other Gram-negative bacilli, and gentamicin sulfate were compared with carbenicillin disodium and gentamicin in a prospective, randomized, double-blind comparison for treating serious Gram-negative infections. Of the 32 patients whose courses were "evaluable" for efficacy, 12 of 14 who received piperacillin and gentamicin and 13 of 18 who received carbenicillin and gentamicin had favorable outcomes. Of the 99 patients whose courses were evaluable for toxicity, nine of 51 recipients of piperacillin and gentamicin and 15 of 48 recipients of carbenicillin and gentamicin suffered clinical reactions possibly, probably, or definitely related to the penicillin. No statistically significant differences were found in the two groups in the frequencies of biochemical abnormalities, including hypokalemia, that occurred in 19 or 44 recipients of piperacillin and gentamicin and 16 of 45 recipients of carbenicillin and gentamicin. Thus, this study did not prove differences in efficacy of toxicity for piperacillin and gentamicin plus carbenicillin and gentamicin for serious Gram-negative infections.
3.Compatibility of verapamil hydrochloride with penicillin admixtures during simulated Y-site injection.
Thompson DF;Stiles ML;Allen LV Jr;Tu YH Am J Hosp Pharm. 1988 Jan;45(1):142-5.
The compatibility of verapamil hydrochloride during simulated Y-site injection with i.v. admixtures containing 11 different penicillins was studied. Admixtures of penicillin G potassium (62.5 mg/mL), nafcillin sodium (40 mg/mL), oxacillin sodium (40 mg/mL), ampicillin sodium (40 mg/mL), carbenicillin disodium (40 mg/mL), methicillin sodium (40 mg/mL), ticarcillin sodium (40 mg/mL), azlocillin sodium (40 mg/mL), mezlocillin sodium (40 mg/mL), piperacillin sodium (40 mg/mL), and amdinocillin (20 mg/mL) were prepared in both 5% dextrose injection and 0.9% sodium chloride injection in minibags. Verapamil hydrochloride injection 4 mL (10 mg) was then added to each admixture, and the admixtures were examined macroscopically and microscopically for precipitate immediately and at 15 minutes and 24 hours after mixing. To simulate Y-site injection of verapamil, verapamil hydrochloride injection 1 mL (2.5 mg) was added to 1 mL of each penicillin admixture in a test tube. For admixtures in which precipitates formed, the pH was recorded before and after verapamil was added to the admixtures. Loss of verapamil hydrochloride when mixed with the penicillin admixtures was determined using reverse-phase high-performance liquid chromatography.
- 相关产品
-
< >
- 推荐产品
-
< >
- 最新产品
-
< >
新闻
怎么做细胞爬片免疫组化染色实验
细胞爬片免疫组化染色,是通过细胞爬片是让玻片浸在细胞培养基内,细胞在玻片上生长,主要用于组织学,免疫组织化学...
2020/7/20 22:04:33
提取病毒RNA的实验方法
提取病毒RNA方法分别有:异硫氰酸胍的提取病毒RNA方法、TRIzol LS提取法、Trizol法提取法等等...
2020/7/22 20:29:26

chelex 100树脂国产替代之路-BIOFOUNT范德生物
Chelex 100螯合离子交换树脂对铜、铁和其他重金属?的偏好显著高于对钠、钾等一价阳离子的偏好。它对二价...
2025/11/4 14:22:46

9月开学季——助研新学期 范德送好礼
2025/8/28 15:30:55

Waxfilm 实验室封口膜:技术与国际市场的双重突破
在实验室耗材领域,封口膜是保障实验准确性与稳定性的关键产品之一。近年来,Waxfilm?实验室封口膜凭借其卓...
2025/5/13 13:03:40

Waxfilm实验室封口膜的5大突破
Waxfilm实验室封口膜作为生物功能膜领域的国产技术突破和品牌突破,是生物领域中国技术发展的缩影。
2025/5/6 17:02:07

各种微流控芯片键合方法的优缺点
微流控芯片键合:目前主要有激光焊接、热压键合、胶键合、超音波焊接,每种方法都有各自的优缺点。本文主要介绍聚酯...
2023/7/28 10:43:09

新一代微流控键合解决方案
微流控键合解决方案:微流控芯片制造的一个重要环节,也是最容易被忽视的--芯片键合。其中一个重要因素是:微流控...
2023/7/27 12:44:28
荧光素钾盐使用说明
D-荧光素钾盐(K+)设计用于体外和体内生物发光测定。D-荧光素的质量和纯度对于获得良好和可重复的结果至关重...
2023/7/20 11:05:11
如何选BSA(牛血清白蛋白)
如何选BSA(牛血清白蛋白):牛血清白蛋白(BSA)有多种形式,如何选择适合自己的牛血清白蛋白(BSA)是一...
2023/2/14 13:09:18




 购物车
购物车 



